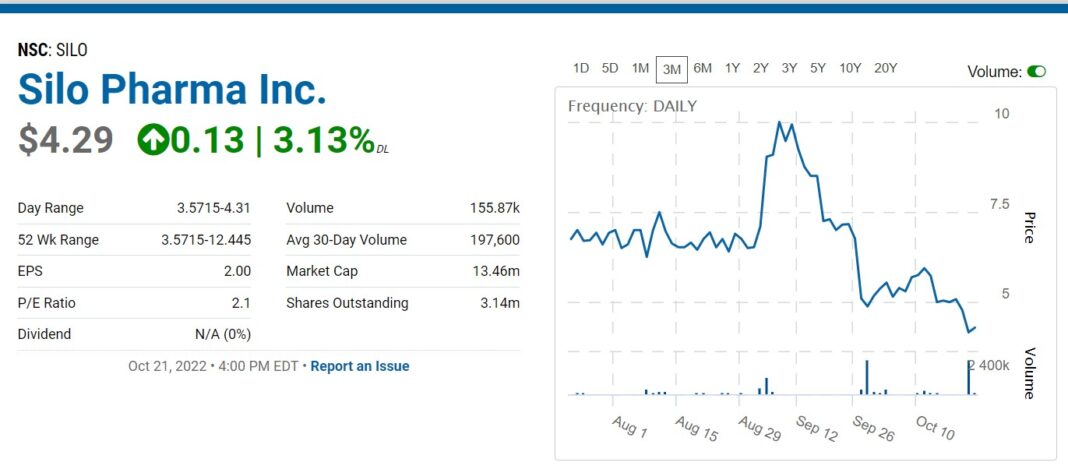
We watched an excellent presentation from management at the Dawson conference and starting our due diligence now. Primary item of our interest is a joint venture with partner Zylö Therapeutics in developing a topical sustained released Ketamine (SP-26) utilizing Z-pod™ technology.
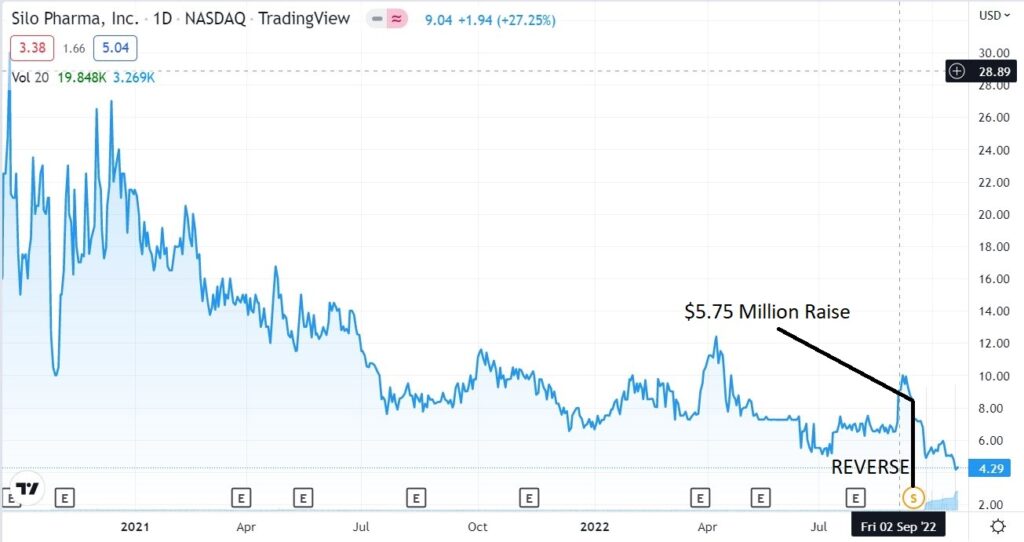
As few investors have heard of Zylö, we could argue this an ‘undiscovered-discovery’ on our part. A good thing in terms of getting involved early, before more investors discover the partnership’s potential. The drop in share price after the recent 50:1 reverse split, adds a double-barreled opportunity in our opinion.
Preclinical studies have already shown that the jointly developed delivery method and formulation can hold and distribute Ketamine in a time-released manner. SP-26 reported positive results in reducing neuropathic nerve pain.
Most importantly is management’s belief that being topical (you rub it on your thigh, versus nasally or intravenously) could lead to patients being able to self-administer at home!
Ketamine as a reminder has been approved nasally for Johnson & Johnson (JNJ) – which bodes well for Silo Pharma – we think you’ll agree.
Self-administered has the potential to reduce the cost of treatment (expanding usage), reduce the complexity of treatment (expanding usage with less Doctor visits) and if used for Fibromyalgia, also expanding usage – as there are few effective treatments for this horrible condition, on the market today (suffered by 4 million US adults). PS, expanding usage can be viewed as the potential for expanding future revenues!!
RELATED: Zylö Therapeutics Closes $5.225 Million Series B Financing.
RELATED: SiloPharma, 50:1 Reverse, Uplist to NASDAQ and $5.75 Million Raise vie Laidlaw (UK).
Silo Pharma Initiates Toxicity Study of its Proprietary Ketamine Formulation for Treatment of Fibromyalgia
Studies of SP-26, the company’s proprietary, time-released ketamine show positive results in reducing neuropathic nerve pain
ENGLEWOOD CLIFFS, NJ, Oct. 10, 2022 (GLOBE NEWSWIRE) — Silo Pharma, Inc. (Nasdaq: SILO) (“the Company”), a developmental stage biopharmaceutical company focused on merging traditional therapeutics with psychedelic research, today announced that it has initiated a preclinical toxicity study of its novel time-released, dosage controlled formulation of ketamine, designated as SP-26, for the treatment of fibromyalgia.
Eric Weisblum, CEO of Silo Pharma, commented, “We are working closely with our joint venture partner Zylö Therapeutics in developing a topical sustained released ketamine utilizing Z-pod™ technology. Preclinical studies have already shown that our joint developed delivery method and formulation can hold and distribute ketamine in a time-released manner. SP-26 reported positive results in reducing neuropathic nerve pain. This safety evaluation study will uncover maximum tolerated dosing data that will inform our future trials.”
In the safety evaluation study, the tolerability of the SP-26 compound will be determined in a mini pig study using an ascending (descending) dosing regimen. Data from the study is expected in the first quarter of 2023.
Silo Pharma recently announced that it is working with a regulatory partner to prepare an FDA Pre-Investigational New Drug (IND) package for SP-26. The Company intends to pursue a 505(b)(2) regulatory pathway for the drug candidate.
About Silo Pharma
Silo Pharma. Inc. is a development-stage biopharmaceutical company focused on merging traditional therapeutics with psychedelic research for people suffering from indications such as PTSD, Alzheimer’s, and other rare neurological disorders. Silo’s mission is to identify assets to license and fund the research which we believe will be transformative to the well-being of patients and the healthcare industry. For more information, visit www.silopharma.com
Forward-Looking Statements
All statements other than statements of historical fact in this announcement are forward-looking statements that involve known and unknown risks and uncertainties and are based on current expectations and projections about future events and financial trends that the Company believes may affect its financial condition, results of operations, business strategy, and financial needs. Investors can identify these forward-looking statements by words or phrases such as “may,” “will,” “expect,” “anticipate,” “aim,” “estimate,” “intend,” “plan,” “believe,” “potential,” “continue,” “is/are likely to” or other similar expressions. The Company undertakes no obligation to update forward-looking statements to reflect subsequent occurring events or circumstances, or changes in its expectations, except as may be required by law. Although the Company believes that the expectations expressed in these forward-looking statements are reasonable, it cannot assure you that such expectations will turn out to be correct, and the Company cautions investors that actual results may differ materially from the anticipated results and encourages investors to review other factors that may affect its future results in the Company’s filings with the SEC.
Contact
800-705-0120
investors@silopharma.com